Laparoscopic right colectomy: technique and atlas
Introduction
The use of laparoscopy in colon surgery quickly evolved since its introduction in the 1990’s from an experimental, infrequently performed approach to the current standard of care (1,2). A laparoscopic approach to right colon resection can be performed for a variety of benign and malignant conditions including resection for ileocolic Crohn’s and colon neoplasia. With careful patient selection and precise operative technique, this approach improves patient recovery using smaller incisions with resulting decrease in patient discomfort and potential shorter hospital stay (3).
Patient selection and workup
When evaluating a patient for potential laparoscopic right colectomy, one must consider potential technical and medical issues that might arise.
A laparoscopic approach will prove difficult in some patients due to technical problems. Specifically, patients who have undergone extensive open abdominal surgery in the past or are clinically obstructed might have an increased risk of bowel injury due to adhesions and poor visualization. In addition, patients with very large masses (malignant or benign) might not be spared a sizeable incision due to the need to remove the surgical specimen.
Patients should be appropriately evaluated preoperatively regarding their cardiopulmonary risk for the use of general anesthesia in this moderate risk operation. Further laboratory assessments should also be performed to assess for anemia, malnutrition or renal dysfunction when appropriate.
Preoperative planning
Preoperative colonoscopy should be reviewed to assess the specific location of the lesion and to ensure that it has been tattooed to aide in identification intraoperatively. The colonoscopy should also be reviewed for concurrent colorectal pathology that may need to be addressed.
Cross-sectional imaging should be reviewed to confirm the location of the lesion as well and assess for tumor extension into adjacent structures (i.e., duodenum), presence of mass effect on the ureter (i.e., hydronephrosis), or evidence of partial obstruction. In non-obstructed patients, preoperative bowel preparation should be performed to reduce the risk of surgical site infection (ref). Removing the column of stool also aides in one’s ability to palpate for lesions and to improve cleanliness during anastomosis of the bowel. For patients who might need a diverting ostomy, preoperative marking and patient education should be performed.
Equipment preference card
- Laparoscopic camera with 10 mm 30-degree scope, light cord, and video display;
- Insufflation tubing and carbon dioxide insufflator;
- 10-mm Hasson port, 5 mm port × 2 (optional 3rd 5 mm port);
- Laparoscopic Bowel graspers;
- 5 mm laparoscopic energy device;
- Wound protector (small);
- Linear stapler.
Procedure
Positioning and port placement
The patient can often be placed in a supine position; however, if the location of the tumor is unclear preoperatively or there is concern for ileosigmoid fistula in a patient with Crohn’s disease, lithotomy position can prove beneficial. Both arms should be tucked at the patient’s side with appropriate padding. In some cases, if the patient is obese, only one arm can be tucked safely. In these instances, the left arm should be tucked preferentially. The hands and fingers should be positioned with the thumbs up in a neutral position to reduce the risk of injury. A foley catheter and orogastric tube should be placed.
The abdomen is prepped and draped after general anesthesia is induced. The operating surgeon stands on the left side of the patient with the assistant on the right for abdominal entry. The assistant then moves to the left side next to the surgeon for the remainder of the operation.
Our preference is to perform a modified Hasson approach for abdominal entry. A 1 cm vertical incision is made and the linea alba is identified. Kocher clamps are used to grasp each side of the midline and cautery is used to open the fascia between. A Kelly clamp is used to open the peritoneum bluntly and a 10 mm port is introduced. Once this is placed, the abdomen is insufflated to 12–15 mmHg with carbon dioxide and a 10 mm scope with a 30-degree angulation is used to survey the abdomen. A 5 mm trocar is placed in the left lower quadrant 2–3 cm medial and superior to the anterior superior iliac spine. This is performed under direct vision with avoidance of the inferior epigastric vessels. A second 5 mm trocar is placed in the left upper quadrant a hands breadth cranially. An additional 5 mm right lower quadrant port is inserted to aide in retraction when needed.
With ports in place, the patient position is adjusted into a 15- to 20-degree tilt with the left side down and slight Trendelenburg. In this position, gravity aides in keeping the small bowel away from the operative field.
Surveillance of the abdomen
The liver, small bowel, large bowel, pelvis and peritoneal surfaces should be assessed for other lesions or tumor implants. Biopsy should be performed and sent for frozen section for suspicious lesions. For patients with Crohn’s disease, the small bowel should be assessed for concurrent areas of disease.
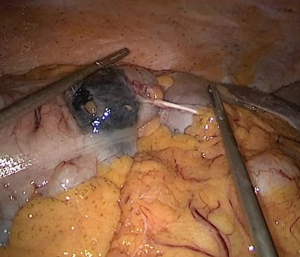
The colonic lesion should next be identified to determine its precise location by observing previously tattooed lesion or palpating the colon with bowel forceps (Figure 1). If the lesion cannot be confidently located, intraoperative colonoscopy with carbon dioxide should be performed. Resection of tumors invading the abdominal wall or adjacent organs should be taken en bloc, and the ability to achieve an R0 resection for malignancy should be determined prior to division of the vasculature.
Identifying the ileocolic pedicle
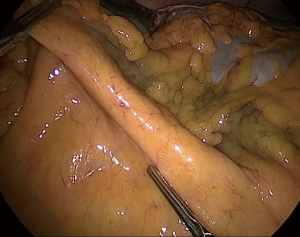
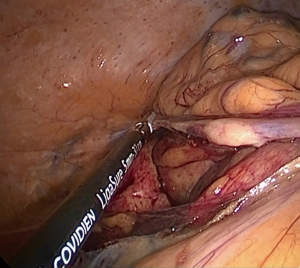
The ileocolic pedicle can be identified by gently placing the ileocecal junction on stretch by pulling it away from the retroperitoneum toward the right lower quadrant using an atraumatic bowel grasper (Figure 2). Once identified, the peritoneum of the mesentery just inferior to the vessel should be opened using the cautery.
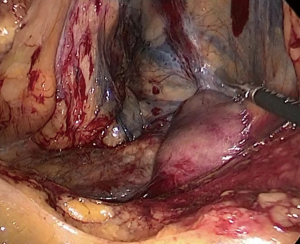
The avascular plane between the mesocolon and retroperitoneum is then developed using blunt dissection, lifting the vessel away from the retroperitoneum. The medial dissection of the artery is carried to its origin from the superior mesenteric artery. Complete dissection in this bloodless plane should be performed laterally over Gerota’s fascia to the abdominal wall laterally and cranially over the duodenum and head of the pancreas entering the lesser sac (Figure 3). Toldt’s fascia should be carefully protected on the retroperitoneum to protect the ureter and duodenum.
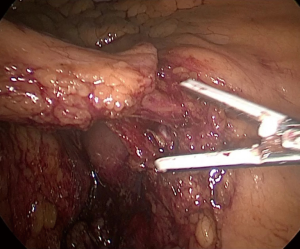
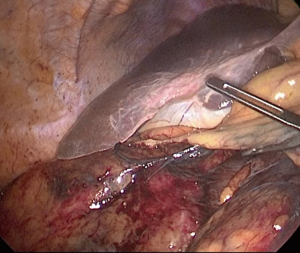
Once the ileocolic artery is isolated it is divided using an energy device, stapler, or surgical clips (Figure 4). Stapling the vessel requires use of a 12-mm left lower quadrant port. While the ileocolic artery is a fairly constant structure, the right colic artery has more variation and can be a branch from the ileocolic or directly off the superior mesenteric artery, which must be transected as well (4,5).
Mobilization of the right colon
After completion of the mesenteric mobilization, the ileal and lateral colonic attachments are divided (Figure 5). The incision should be made where the peritoneal reflection meets the lateral bowel wall starting at the cecum and moving cranially to the hepatic flexure. An atraumatic bowel grasper is used to pull the right colon medially and lift it off the retroperitoneum to provide traction while this lateral dissection is carried out. The area of prior dissection will be entered and the dissection is carried up to and around the hepatic flexure. The hepatocolic ligament is divided using cautery, taking care not to cause injury to the nearby gallbladder and duodenum (Figure 6). Finally, the greater omentum is divided from the transverse colon and the gastrocolic ligament is divided. At the end of the dissection the right colon should be completely mobile and easily medialized. If dissection of the hepatic flexure is difficult, moving the patient into reverse Trendelenburg or inserting a right upper quadrant port for additional traction is sometimes necessary (6).
The attachments tethering the ileum to the retroperitoneum must be taken down to allow complete mobility of the bowel. Scissors also should be used when taking down these thin attachments moving cephalad to avoid injury to the third portion of the duodenum and superior mesenteric artery origin.
Transverse colon mesentery dissection
The right branches of the middle colic vessels should be identified and divided next with surgical clips or an energy device. This division is necessary to aide in colon mobility for eventual evisceration of the bowel for specimen removal and for anastomosis.
Special considerations
When planning the extent of right colon resection and level of vascular ligation one must consider the underlying pathology. When the operation is performed for malignancy the vessels should be ligated high at their origin to ensure an adequate lymphadenectomy and care should be taken to keep the mesocolon intact (5,7-10). In addition, a “no touch technique” should be employed for cases of malignancy to avoid manipulation of the tumor itself. In contrast, high ligation of the vasculature adds unnecessary risk when the operation is performed for benign disease.
Extracorporeal anastomosis
With the mesentery divided and mobilization complete, the bowel should be grasped with a ratcheted grasper, pneumoperitoneum relieved, and the umbilical port removed. The umbilical incision should be extended vertically to approximately 4 cm to allow evisceration of the bowel. This might need to be elongated if there is a phlegmon or a large mass. A wound protector is placed at this time. The right colon is grasped using a Babcock clamp externally and exteriorized. Care should be taken to ensure that the mesentery is not twisted when eviscerating the bowel.
The terminal ileum is evaluated and divided at a site of healthy, well-perfused bowel. A linear cutting stapler is used to divide the bowel. The transverse colon is then inspected. The site of division should occur where the bowel is healthy and well perfused. Division of the mesentery and resulting pulsatile bleeding can confirm good perfusion to the segment. With good hemostasis a linear cutting stapler can be used to divide the transverse colon. The specimen is then removed from the field and examined to confirm that the pathology of interest is included in the specimen.
Prior to performing an anastomosis both ends of the bowel should be assessed again for viability. Either a hand-sewn or stapled anastomosis may be performed. Our preference is to perform a stapled anastomosis in most situations. A hand sewn technique may be considered in situations where staplers may be ill-advised, such as poor tissues in Crohn’s disease. Enterotomies are made on the antimesenteric ends of the ileum and transverse colon and a linear cutting stapler [i.e., gastrointestinal anastomosis (GIA)] is placed inside to align them in a side-to-side fashion. The linear stapler is inserted, ensuring that the mesentery is not caught in the staple line during alignment. After the first firing of the stapler, the bowel lumen is assessed for bleeding. The bowel opening is then aligned to mismatch the first staple firing and a second transverse anastomosis (TA) linear stapler is used to close the ends of the bowel (an additional GIA linear staple load may be used). The staple lines are inspected for hemostasis and 3-0 absorbable suture is used to reinforce the staple line ends. An apex (“crotch”) stitch is placed to help relieve some tension off the anastomosis. If available, omentum can be used to cover the anastomosis. The mesenteric window is not closed, and the bowel is carefully reduced into the abdomen.
Intracorporeal anastomosis
After transection of the vessels and mobilization of the bowel, the transverse colon and terminal ileum are transected using the laparoscopic linear cutting stapler. The specimen is placed in an endocatch bag over the liver to maximize working space. The transverse colon and ileum are then aligned in a side to side fashion and a stay suture is placed on the antimesenteric border about 15 cm away from the stapled ends. Enterotomies are made in the antimesenteric border of each piece of bowel near the staple line using the cautery. A second stay suture is then placed at the enterotomy site closest to the staple line. A linear cutting stapler is then fed into the enterotomy carefully, ensuring that the mesentery is free underneath. This stapler is fired to create the side to side anastomosis. A free suture is then used to suture the enterotomy site closed in a running fashion. A second continuous suture line is then performed in a running fashion using seromuscular bites using the stay sutures to help manipulate the bowel. The umbilical incision is then extended to extract the specimen (11-13).
Role of team members
It is important to ensure that all members of the surgical team are focused on the safety of the patient during the operation. Proper patient positioning and securing with assistance from the anesthesia and nursing staff can decrease the risk of nerve injury and malpositioning when the patient position is changed during the operation to improve visualization. Conversion to an open procedure if a problem is encounter, such as uncontrolled hemorrhage or intolerance of insufflation with bradycardia or poor ventilation, is an important and safe decision if needed.
Postoperative management
For elective right colectomy our patients are provided a liquid diet on POD0 with advancement to a soft diet the following morning. Our institution uses a bundled care enhanced recovery plan to aide in the minimization of narcotic use, support goal directed fluid therapy, and allow early mobilization, which has resulted in a shortened hospital stay.
Tips, Tricks and Pitfalls
- When performing medial mobilization of the ileocolic artery for cancer, the initial peritoneal incision should be close to the superior mesenteric artery (SMA) to ensure a high ligation of the vessel;
- If Toldt’s fascia is carefully protected over the retroperitoneum, it will avoid injury to the ureter, the duodenum, and the retroperitoneal structures;
- When providing traction, remember to pull the colon off the retroperitoneal structures to aide in visualizing the planes;
- Mobilizing the greater omentum and transverse colon to or beyond the midline facilitates specimen extraction, particularly in more obese individuals;
- For obese individuals, moving the left sided ports closer to the midline will aide in one’s ability to reach the right upper quadrant for dissection of the hepatic flexure.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Michael J Stamos and Mehraneh Dorna Jafari) for the series “Laparoscopic Colon Surgery” published in Annals of Laparoscopic and Endoscopic Surgery. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/ales.2019.01.01). The series “Laparoscopic Colon Surgery” was commissioned by the editorial office without any funding or sponsorship. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Jacobs M, Verdeja JC, Goldstein HS. Minimally invasive colon resection (laparoscopic colectomy). Surg Laparosc Endosc 1991;1:144-50. [PubMed]
- Falk PM, Beart RW Jr, Wexner SD, et al. Laparoscopic colectomy: a critical appraisal. Dis Colon Rectum 1993;36:28-34. [Crossref] [PubMed]
- Ding J, Liao GQ, Xia Y, et al. Laparoscopic versus open right hemicolectomy for colon cancer: a meta-analysis. J Laparoendosc Adv Surg Tech A 2013;23:8-16. [Crossref] [PubMed]
- Delaney C. Netter's Surgical Anatomy and Approaches. Philadelphia: Saunders, 2013.
- Alsabilah JF, Razvi SA, Albandar MH, et al. Intraoperative Archive of Right Colonic Vascular Variability Aids Central Vascular Ligation and Redefines Gastrocolic Trunk of Henle Variants. Dis Colon Rectum 2017;60:22-9. [Crossref] [PubMed]
- Delaney CP. Operative Techniques in Laparoscopic Colorectal Surgery. Second edition. Philadelphia, PA: Lippincott Williams & Wilkins, 2014.
- Adamina M, Manwaring ML, Park KJ, et al. Laparoscopic complete mesocolic excision for right colon cancer. Surg Endosc 2012;26:2976-80. [Crossref] [PubMed]
- Siani LM, Lucchi A, Berti P, et al. Laparoscopic complete mesocolic excision with central vascular ligation in 600 right total mesocolectomies: Safety, prognostic factors and oncologic outcome. Am J Surg 2017;214:222-7. [Crossref] [PubMed]
- Lorenzon L, Biondi A, Carus T, et al. Achieving high quality standards in laparoscopic colon resection for cancer: A Delphi consensus-based position paper. Eur J Surg Oncol 2018;44:469-83. [Crossref] [PubMed]
- Ye K, Lin J, Sun Y, et al. Variation and treatment of vessels in laparoscopic right hemicolectomy. Surg Endosc 2018;32:1583-4. [Crossref] [PubMed]
- Rossini R, Lisi G, Gentile I, et al. Intracorporeal Ileocolic Anastomosis in Laparoscopic Right Colectomy: A New Way to Make it Simple? Surg Technol Int 2018;32:115-8. [PubMed]
- Jian-Cheng T, Shu-Sheng W, Bo Z, et al. Total laparoscopic right hemicolectomy with 3-step stapled intracorporeal isoperistaltic ileocolic anastomosis for colon cancer: An evaluation of short-term outcomes. Medicine (Baltimore) 2016;95:e5538 [Crossref] [PubMed]
- Abrisqueta J, Ibanez N, Lujan J, et al. Intracorporeal ileocolic anastomosis in patients with laparoscopic right hemicolectomy. Surg Endosc 2016;30:65-72. [Crossref] [PubMed]
Cite this article as: Cauley CE, Valente MA, Champagne BJ. Laparoscopic right colectomy: technique and atlas. Ann Laparosc Endosc Surg 2019;4:15.