Observing an upward trajectory in minimally invasive hepatectomies in Singapore—a nationwide analysis
Introduction
Laparoscopy was first introduced as a diagnostic tool in the 1960s, with the first laparoscopic cholecystectomy being performed in 1987. Since then, laparoscopic abdominal surgery has advanced rapidly to include major and complex oncological resections (1). While it has been adopted widely adopted across other major abdominal organ systems, laparoscopy’s application in hepatobiliary and pancreatic surgery has only started to see widespread enthusiasm in recent years. A sharp upward trajectory has been observed in minimally invasive hepatectomies (MIH) or laparoscopic liver resections (LLR).
Since the first reported case of MIH for a benign liver lesion in 1991 performed with a fully laparoscopic approach (LLR) (2), MIH have become commonplace in liver resections for both benign and malignant disease and is now also deployed in living related donor hepatectomies. The first International Consensus Conference on Laparoscopic Liver Surgery held in Louisville in 2008 determined that MIH was safe and effective in selected patients but recognized that there was a significant learning curve as a result of the technical difficulties in LLRs, especially that seen in major resections (3). The second International Consensus Conference held in Morioka, Japan in 2014 further defined the role of MIH and LLR, specifically the role of other hybrid techniques such as hand-assisted laparoscopic surgery (HALS) (4). It also further clarified the role of major robotic surgery and laparoscopic donor hepatectomies. The committee found a significant global spread of MIH to have occurred after the first International Consensus meeting and with it, an associated decrease in conversion rates. While minor MIH continues to contribute the majority of cases of LLR worldwide, there has been a significant increase in the proportion of major and anatomic MIH being performed globally in recent years. Current available reports suggest that the most significant increases in MIH activities have taken place in East Asia, North America and Europe (5,6).
The benefits of LLR and other minimally invasive hybrid methods are undisputed. Well documented benefits of MIH include reduced intra-operative blood loss and thus less need for blood transfusions, smaller surgical incisions with less incision-related complications, shorter duration of hospitalization and overall enhanced post-operative recovery (7-11). There is a substantial learning curve in MIH at an individual as well as at institutional level before attaining competency and expertise (12-18). As such, early adoption of MIH with the gradual introduction of increasing difficulty will assist in scaling the learning curve proficiently. A novel difficulty scoring for LLR could potentially help in shortening the learning curve by predicting procedure difficulty according to Ban et al. (19,20).
Since 2004, MIH has been gradually adopted in the major public hospitals in Singapore and we have seen a rapid upward trajectory of its implementation in the past few years. In this study, we describe the trends of minimally invasive hepatectomies in Singapore.
Methods
A retrospective review of all cases of MIH from 4 major centers performing MIH [Singapore General Hospital (SGH), National University Hospital (NUH), Changi General Hospital (CGH) and Ng Teng Fong General Hospital (NTFGH)] between December 2004 and February 2018 was performed. Data from CGH and NTFGH were collected from the start of their MIH program in 2014 and 2016 respectively. Historical, demographic and disease related data including the indication for surgery was collected. Outcome data including the length of hospital stay (LOS), morbidity and mortality was also collected and analyzed. An analysis of case distribution between the 4 hospitals and trends in case numbers was performed.
Statistical analysis was performed using IBM SPSS software version 23 (SPSS, Chicago, IL, USA). Continuous data was expressed in median values (range) while categorical data was expressed as number with percentages.
Results
The first case of MIH in Singapore was performed at the SGH in 2004. The case was a minor wedge resection of an 18 mm tumour in segment 5 of the liver for diagnostic purposes which turned out to be a poorly differentiated metastatic nodule. Subsequently, the first major MIH was performed at the NUH in 2006, in which a full laparoscopic right hepatectomy was performed for hepatocellular carcinoma (HCC). The operative time was 420 minutes with a total LOS of 8 days. There was no morbidity or mortality for both of these early cases in Singapore. Since then, two other centers (CGH and NTFGH) have begun their MIH programs.
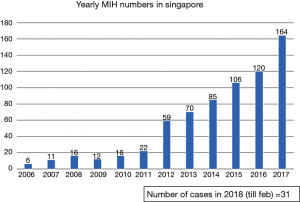
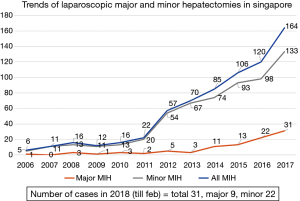
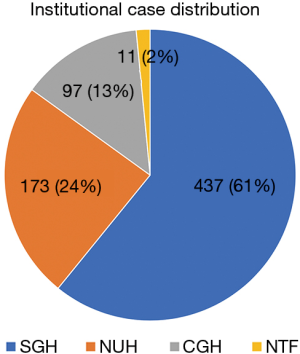
A total of 718 cases across the four institutions from December 2004 to February 2018 were analyzed. The increasing trend of MIH cases over the years is shown in Figure 1. The inflection points of the upward trajectory took place in year 2012. In addition to the gradual increase in the absolute number of cases on a yearly basis, we also observed an increase in the proportion of major MIH annually as shown in Figure 2. When reviewing all the MIH cases, major MIH constituted 14% (n=101) while minor resection accounted for the remaining 86% of LLR cases (n=617). The distribution of cases across the various institutions is shown in Figure 3.
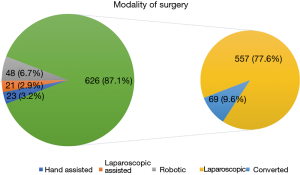
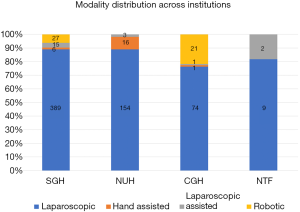
The most common modality of surgery was full laparoscopic LLR (n=626, 87.1%) followed by robotic MIH (n=48, 6.7%). Hand-assisted approach (n=23, 3.2%) and laparoscopic-assisted approach (n=21, 2.9%) were less commonly performed (Figure 4). The distribution of various surgical modalities across the 4 institutions is shown in Figure 5. All cases of robotic surgery were performed at SGH and CGH. In the laparoscopic group, the overall rate of conversion was 9.6% (n=69). Of the conversions, 94.2% (n=65) were converted to an open approach, while 5.8% (n=4) were converted to a hand assisted approach.
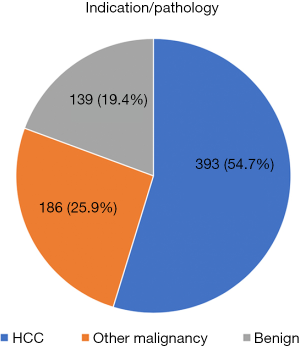
The most common pathology requiring surgery was HCC which accounted for 54.7% of cases (n=393), followed by other liver malignancies including colorectal liver metastases and intrahepatic cholangiocarcinoma (25.9%, n=186) and benign liver lesions (19.4%, n=139) as shown in Figure 6. The median operating time was 245 (30–887) minutes, with a median blood loss of 375 (0–5,000) mLs. The median length of stay was 4 days, with a maximum reported length of 79 days. The overall 30- and 90-day mortality was 0.3% and 0.7% respectively. Rate of major morbidity (Clavien grade 3 or more) was 5.4% (n=39). Data can be found in Table 1.
Table 1
| Operative metrics and overall clinical outcomes | Values |
|---|---|
| Median duration of surgery (min) | 245 (30–887) |
| Median estimated blood loss (mLs) | 375 (0–5,000) |
| Median LOS (days) | 4 (1–79) |
| 30-day mortality | 2 (0.3%) |
| 90-day mortality | 5 (0.7%) |
| Major morbidity (Clavien >2) | 39 (5.4%) |
| Conversion | 69 (9.6%) |
LOS, length of stay.
Discussion
The laparoscopic approach to major abdominal surgery has gained worldwide acceptance in the last 3 decades. This has mainly been driven by improved patient selection coupled with new advances in laparoscopic instruments, including laparoscopic energy devices, stapling technologies and other advances that are essential in performing complex abdominal operations safely. While the cost effectiveness of MIH was an early concern in its implementation, recent data published including the OSLO-Comet study in colorectal liver metastases have demonstrated that MIH is a cost-effective method of performing liver resections (21). We currently await further evidence confirming the other benefits of MIH, especially since the premature closure of the ORANGE-II trial due to slow patient recruitment (22).
The adoption of laparoscopy in major liver resections has progressed markedly slower in comparison to other surgical disciplines such as that in gynaecological and colorectal surgery. The lack of high-level evidence supporting its benefit over open resections is likely one of the first major hurdles. Other possible reasons for slow adoption of MIH include the technical complexity of liver surgery, the large variation in technical difficulty across different types of resections depending on the location of the lesion(s) and the proximity of the hepatic lesions to major vascular and biliary structures. Hence, surgeons generally adopt a graduated approach to MIH where a concerted effort is made in selecting cases of gradually increasing complexity and difficulty—such as progressing from a simple wedge resection in peripheral segments to laparoscopic left lateral sectionectomy and subsequently to major resections to ensure patient safety while allowing the operating surgeon to gain competence (12,18). While the consensus conference in 2014 declared major hemihepatectomies still as an innovative procedure (4), the application of laparoscopy to major resections has become widely practiced in Singapore. The highest proportion of major MIH is seen in NUH with 24.3% of the cases (42 out of 173 cases) being major MIH. A recent large series by van der Poel et al suggested a learning curve of 55 cases for major hemihepatectomies using a risk-adjusted cumulative sum analysis for conversion (23). Beyond that, the number of more complex LLRs including central hepatectomies and extended lobectomies or trisectionectomies has also increased gradually since 2015 with the first laparoscopic central hepatectomy and extended left hepatectomy performed in SGH. The first laparoscopic associating-liver partition and portal ligation (ALPSS) was performed in NUH in 2017.
The increasing number of major resections in each institution in our series has demonstrated the gradual learning curve and experience collectively, with a sharp upward trajectory observed in 2012. The MIH program the CGH started in Nov 2014 with a rapid increase in cases since, benefiting from the pioneering work from NUH and SGH and shared experience and collaboration across public institutions in the country. NTF subsequently followed in 2016 after its opening in 2015. Goh et al. from the SGH reported a gradual decrease in conversion over the years in their series of 400 cases of MIH, with an increasing proportion of major resections as well as the proportion of resections in posterosuperior locations (24). Concurrent data from NUH has demonstrated that despite an increase in the median difficulty scoring system (DSS) scores over sequential 5-year periods from 2006–2017, there has been a significant decrease in conversion rates. As each institution, and the country as a whole, gains more experience with MIH we anticipate a further increase in major resections across all institutions in the coming years.
The most common disease for which MIH was performed in our series was HCC. A report from NUH previously reported their results of MIH, showing reduced blood loss, blood transfusions and shorter hospital LOS in the MIH group compared to open resections for HCC (25). However, the same series suggested that the short-term benefits might have come at the expense of a shorter disease-free interval. Since then however, larger studies including a systematic review by Goh et al. have demonstrated the oncological equivalence of MIH compared to Open hepatectomy (7,26-28). Similarly, in colorectal cancer liver metastases, which was the second most common indication for surgery, the short-term benefits of MIH have been repeatedly demonstrated (29). While high level evidence is still lacking, a number of retrospective series have shown MIH to have comparable oncological outcomes to open hepatectomy (30-33). In the Oslo-COMET trial, rates of R1 resections were comparable between both arms (21). We await the results of the ongoing ORANGE-II PLUS trial in supporting MIH in hemihepatectomies.
The limited visualization associated with a lack of articulated instruments has prompted an enthusiasm in robotic assistance in hepatic resections, especially given the technical demands in more complex cases. While still considered developmental, recent reports have demonstrated the safety and feasibility of robotic liver surgery (RLS) both in terms of perioperative outcomes and long-term oncological outcomes (34,35). Furthermore, the use of a robotic platform has been reported to facilitate MIH in cases which might be presumed to be too complex for a pure laparoscopic approach. Wu et al. reported an increase in successful MIH from 15% to 44% with robotic assistance (36). SGH has performed the largest number of RLS for liver resection followed closely by CGH, which has utilized RLS early in their learning curve to facilitate their MIH program. In this way, RLS can be used to facilitate complex MIH early in a center’s learning curve.
The evidence to date suggests comparable outcomes with RLS compared to LLR in terms of LOS, R0 resection rate, operative time, morbidity and mortality (37,38). While certain steps such as retrohepatic caval dissection, portal dissection and in approaching posterosuperior segments might be facilitated by the robotic platform, whether this translates to a difference in clinical outcomes remains to be seen. Locally, the RLS platform has been used as an extension of minimally invasive hepatectomy complementary to a laparoscopic approach. In their early experience, non-anatomical resections in the difficulty segments have been facilitated greatly with the RLS platform. The combined early experience in CGH and SGH showed that RLS is a safe and feasible minimally invasive technique that can be applied to complex resections such as that seen in major hepatectomies, resections in posterosuperior segments and repeat liver resections with low morbidity and low open conversion rates (39). Despite that, given Singapore’s publicly funded healthcare system, the costs with RLS remains a concern.
While donor safety is of paramount importance in living donor liver transplantation (LDLT), the hope of reducing incisions/scars while improving postoperative recovery has motivated the application of minimally invasive surgical techniques to donor hepatectomies. Since the first laparoscopic donor left lateral sectionectomy (LLS) in 2002 (40), the experience especially in East Asia has led to tremendous increases in minimally invasive donor hepatectomies. Laparoscopic donor LLS has now become commonplace with multiple series reporting its safety, with an expert panel in 2017 declaring it standard practice in pediatric LDLT (41-43). The first right lobe donor hepatectomy was subsequently performed in 2013 (44). With increasing experience, laparoscopic right lobe donor hepatectomies have become commonplace in high volume centres, with a recent report of 115 cases from Lee et al. showing low morbidity and no mortalities (45,46). In their series there were clear differences in outcomes with increasing experience, with the authors suggesting a learning curve of approximately 60 cases a year. Since November 2017, 3 cases of totally laparoscopic LLS living donor harvesting have been performed in NUH without major morbidity or mortality, with one case requiring graft size reduction intracorporeally as the recipient was a very small baby. The mean operative time was 372 minutes. All patients were discharged well.
Conclusions
We believe in the benefits of MIH over open resections in terms of short and long-term patient and oncological outcomes. The implementation of a MIH program in any institution requires meticulous advance planning and a concerted effort to build capabilities without compromising patient safety. While we await further high-level evidence supporting MIH implementation in the various fields of oncological surgery and transplantation, we will continue to build each institution’s MIH capability as a country.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, Annals of Laparoscopic and Endoscopic Surgery for the series “Advanced Laparoscopic HPB Surgery in South East Asia”. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/ales.2018.08.05). The series “Advanced Laparoscopic HPB Surgery in South East Asia” was commissioned by the editorial office without any funding or sponsorship. Chiow KH, Lee SY and Chan CY served as the unpaid Guest Editors of the series. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). Data collected across all institutions was performed with the ethical approval of the respective institutional review boards (IRB). Individual consent for this retrospective analysis was waived.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Litynski GS. Profiles in laparoscopy: Mouret, Dubois, and Perissat: the laparoscopic breakthrough in Europe (1987-1988). JSLS 1999;3:163-7. [PubMed]
- Reich H, McGlynn F, DeCaprio J, et al. Laparoscopic excision of benign liver lesions. Obstet Gynecol 1991;78:956-8. [PubMed]
- Buell JF, Cherqui D, Geller DA, et al. The international position on laparoscopic liver surgery: The Louisville Statement, 2008. Ann Surg 2009;250:825-30. [Crossref] [PubMed]
- Wakabayashi G, Cherqui D, Geller DA, et al. Recommendations for laparoscopic liver resection: a report from the second international consensus conference held in Morioka. Ann Surg 2015;261:619-29. [PubMed]
- Hibi T, Cherqui D, Geller DA, et al. Expanding indications and regional diversity in laparoscopic liver resection unveiled by the International Survey on Technical Aspects of Laparoscopic Liver Resection (INSTALL) study. Surg Endosc 2016;30:2975-83. [Crossref] [PubMed]
- Hibi T, Cherqui D, Geller DA, et al. International Survey on Technical Aspects of Laparoscopic Liver Resection: a web-based study on the global diffusion of laparoscopic liver surgery prior to the 2nd International Consensus Conference on Laparoscopic Liver Resection in Iwate, Japan. J Hepatobiliary Pancreat Sci 2014;21:737-44. [Crossref] [PubMed]
- Takahara T, Wakabayashi G, Beppu T, et al. Long-term and perioperative outcomes of laparoscopic versus open liver resection for hepatocellular carcinoma with propensity score matching: a multi-institutional Japanese study. J Hepatobiliary Pancreat Sci 2015;22:721-7. [Crossref] [PubMed]
- Wu X, Huang Z, Lau WY, et al. Perioperative and long-term outcomes of laparoscopic versus open liver resection for hepatocellular carcinoma with well-preserved liver function and cirrhotic background: a propensity score matching study. Surg Endosc 2018; [Epub ahead of print]. [Crossref] [PubMed]
- Ciria R, Cherqui D, Geller DA, et al. Comparative Short-term Benefits of Laparoscopic Liver Resection: 9000 Cases and Climbing. Ann Surg 2016;263:761-77. [Crossref] [PubMed]
- Ocuin LM, Tsung A. Minimally Invasive Hepatic Surgery. Surg Clin North Am 2016;96:299-313. [Crossref] [PubMed]
- Yan Y, Cai X, Geller DA. Laparoscopic Liver Resection: A Review of Current Status. J Laparoendosc Adv Surg Tech A 2017;27:481-6. [Crossref] [PubMed]
- Komatsu S, Scatton O, Goumard C, et al. Development Process and Technical Aspects of Laparoscopic Hepatectomy: Learning Curve Based on 15 Years of Experience. J Am Coll Surg 2017;224:841-50. [Crossref] [PubMed]
- Kluger MD, Vigano L, Barroso R, et al. The learning curve in laparoscopic major liver resection. J Hepatobiliary Pancreat Sci 2013;20:131-6. [Crossref] [PubMed]
- Nomi T, Fuks D, Kawaguchi Y, et al. Learning curve for laparoscopic major hepatectomy. Br J Surg 2015;102:796-804. [Crossref] [PubMed]
- Cheek SM, Geller DA. The Learning Curve in Laparoscopic Major Hepatectomy: What Is the Magic Number? JAMA Surg 2016;151:929. [Crossref] [PubMed]
- Chan FK, Cheng KC, Yeung YP, et al. Learning Curve for Laparoscopic Major Hepatectomy: Use of the Cumulative Sum Method. Surg Laparosc Endosc Percutan Tech 2016;26:e41-5. [Crossref] [PubMed]
- Brown KM, Geller DA. What is the Learning Curve for Laparoscopic Major Hepatectomy? J Gastrointest Surg 2016;20:1065-71. [Crossref] [PubMed]
- Goh BK, Teo JY, Lee SY, et al. Critical appraisal of the impact of individual surgeon experience on the outcomes of laparoscopic liver resection in the modern era: collective experience of multiple surgeons at a single institution with 324 consecutive cases. Surg Endosc 2018;32:1802-11. [Crossref] [PubMed]
- Hasegawa Y, Wakabayashi G, Nitta H, et al. A novel model for prediction of pure laparoscopic liver resection surgical difficulty. Surg Endosc 2017;31:5356-63. [Crossref] [PubMed]
- Im C, Cho JY, Han HS, et al. Validation of difficulty scoring system for laparoscopic liver resection in patients who underwent laparoscopic left lateral sectionectomy. Surg Endosc 2017;31:430-6. [Crossref] [PubMed]
- Fretland AA, Dagenborg VJ, Bjornelv GM, et al. Laparoscopic Versus Open Resection for Colorectal Liver Metastases: The OSLO-COMET Randomized Controlled Trial. Ann Surg 2018;267:199-207. [Crossref] [PubMed]
- Wong-Lun-Hing EM, van Dam RM, van Breukelen GJ, et al. Randomized clinical trial of open versus laparoscopic left lateral hepatic sectionectomy within an enhanced recovery after surgery programme (ORANGE II study). Br J Surg 2017;104:525-35. [Crossref] [PubMed]
- van der Poel MJ, Besselink MG, Cipriani F, et al. Outcome and Learning Curve in 159 Consecutive Patients Undergoing Total Laparoscopic Hemihepatectomy. JAMA Surg 2016;151:923-8. [Crossref] [PubMed]
- Goh BK, Lee SY, Teo JY, et al. Changing trends and outcomes associated with the adoption of minimally invasive hepatectomy: a contemporary single-institution experience with 400 consecutive resections. Surg Endosc 2018; [Epub ahead of print]. [Crossref] [PubMed]
- Leong WQ, Ganpathi IS, Kow AW, et al. Comparative study and systematic review of laparoscopic liver resection for hepatocellular carcinoma. World J Hepatol 2015;7:2765-73. [Crossref] [PubMed]
- Kasai M, Cipriani F, Gayet B, et al. Laparoscopic versus open major hepatectomy: a systematic review and meta-analysis of individual patient data. Surgery 2018;163:985-95. [Crossref] [PubMed]
- Goh EL, Chidambaram S, Ma S. Laparoscopic vs open hepatectomy for hepatocellular carcinoma in patients with cirrhosis: A meta-analysis of the long-term survival outcomes. Int J Surg 2018;50:35-42. [Crossref] [PubMed]
- Kang WH, Kim KH, Jung DH, et al. Long-term results of laparoscopic liver resection for the primary treatment of hepatocellular carcinoma: role of the surgeon in anatomical resection. Surg Endosc 2018; [Epub ahead of print]. [Crossref] [PubMed]
- Hallet J, Sa Cunha A, Cherqui D, et al. Laparoscopic Compared to Open Repeat Hepatectomy for Colorectal Liver Metastases: a Multi-institutional Propensity-Matched Analysis of Short- and Long-Term Outcomes. World J Surg 2017;41:3189-98. [Crossref] [PubMed]
- Allard MA, Cunha AS, Gayet B, et al. Early and Long-term Oncological Outcomes After Laparoscopic Resection for Colorectal Liver Metastases: A Propensity Score-based Analysis. Ann Surg 2015;262:794-802. [Crossref] [PubMed]
- Untereiner X, Cagniet A, Memeo R, et al. Laparoscopic hepatectomy versus open hepatectomy for colorectal cancer liver metastases: comparative study with propensity score matching. Hepatobiliary Surg Nutr 2016;5:290-9. [Crossref] [PubMed]
- Lewin JW, O’Rourke NA, Chiow AK, et al. Long-term survival in laparoscopic vs open resection for colorectal liver metastases: inverse probability of treatment weighting using propensity scores. HPB (Oxford) 2016;18:183-91. [Crossref] [PubMed]
- Martinez-Cecilia D, Cipriani F, Vishal S, et al. Laparoscopic Versus Open Liver Resection for Colorectal Metastases in Elderly and Octogenarian Patients: A Multicenter Propensity Score Based Analysis of Short- and Long-term Outcomes. Ann Surg 2017;265:1192-200. [Crossref] [PubMed]
- Wong DJ, Wong MJ, Choi GH, et al. Systematic review and meta-analysis of robotic versus open hepatectomy. ANZ J Surg 2018; [Epub ahead of print]. [Crossref] [PubMed]
- Khan S, Beard RE, Kingham PT, et al. Long-Term Oncologic Outcomes Following Robotic Liver Resections for Primary Hepatobiliary Malignancies: A Multicenter Study. Ann Surg Oncol 2018; [Epub ahead of print]. [Crossref] [PubMed]
- Wu YM, Hu RH, Lai HS, et al. Robotic-assisted minimally invasive liver resection. Asian J Surg 2014;37:53-7. [Crossref] [PubMed]
- Hanna T, Imber C. Robotics in HPB surgery. Ann R Coll Surg Engl 2018;100:31-7. [Crossref] [PubMed]
- Ocuin LM, Tsung A. Robotic liver resection for malignancy: Current status, oncologic outcomes, comparison to laparoscopy, and future applications. J Surg Oncol 2015;112:295-301. [Crossref] [PubMed]
- Goh BK, Lee LS, Lee SY, et al. Initial experience with robotic hepatectomy in Singapore: analysis of 48 resections in 43 consecutive patients. ANZ J Surg 2018; [Epub ahead of print]. [Crossref] [PubMed]
- Cherqui D, Soubrane O, Husson E, et al. Laparoscopic living donor hepatectomy for liver transplantation in children. Lancet 2002;359:392-6. [Crossref] [PubMed]
- Han HS, Cho JY, Kaneko H, et al. Expert Panel Statement on Laparoscopic Living Donor Hepatectomy. Dig Surg 2018;35:284-8. [Crossref] [PubMed]
- Soubrane O, de Rougemont O, Kim KH, et al. Laparoscopic Living Donor Left Lateral Sectionectomy: A New Standard Practice for Donor Hepatectomy. Ann Surg 2015;262:757-61; discussion 761-3. [Crossref] [PubMed]
- Broering DC, Elsheikh Y, Shagrani M, et al. Pure Laparoscopic Living Donor Left Lateral Sectionectomy in Pediatric Transplantation: A Propensity Score Analysis on 220 Consecutive Patients. Liver Transpl 2018;24:1019-30. [Crossref] [PubMed]
- Soubrane O, Perdigao Cotta F, Scatton O. Pure laparoscopic right hepatectomy in a living donor. Am J Transplant 2013;13:2467-71. [Crossref] [PubMed]
- Suh KS, Hong SK, Lee KW, et al. Pure laparoscopic living donor hepatectomy: Focus on 55 donors undergoing right hepatectomy. Am J Transplant 2018;18:434-43. [Crossref] [PubMed]
- Lee KW, Hong SK, Suh KS, et al. One Hundred and Fifteen Cases of Pure Laparoscopic Living Donor Right Hepatectomy at a Single Center. Transplantation 2018; [Epub ahead of print]. [PubMed]
Cite this article as: Tan CHN, Tan KHJ, Lee CL, Lee LS, Chiow KHA, Goh BKP, Lee SY, Chan CY, Ganpathi IS, Kow WCA. Observing an upward trajectory in minimally invasive hepatectomies in Singapore—a nationwide analysis. Ann Laparosc Endosc Surg 2018;3:78.