
Secrets for a successful laparoscopic antireflux surgery: patients with extraesophageal symptoms
Introduction
The Montreal definition and classification of gastro esophageal reflux disease (GERD) defined GERD simply as a condition that develops when reflux of gastric contents causes troublesome symptoms or complications (1). This consensus pointed out extraesophageal manifestations of GERD, including cough, laryngitis, asthma and dental erosion (1).
Two mechanisms are proposed to explain extraesophageal GERD syndromes. The first is a direct reflux to the target organ with irritation or microaspiration and the second a reflex (2-4) inciting an esophagus-tracheobronchial reflex that is mediated by the vagus nerve and triggered by chemical stimulation of the esophagus (5,6).
The same Montreal consensus pointed that “medical and surgical treatment trials aimed at improving presumed reflux cough, reflux laryngitis, and reflux asthma syndromes by treating GERD are associated with uncertain and inconsistent treatment effect”. Indeed, some studies showed worse outcomes for patients with extra compared to esophageal symptoms after surgical therapy (7,8). This may be explained by the fact that a cause-and-effect relationship between GERD and extra esophageal symptoms is often difficult to establish since other disorders of the lung and upper respiratory tract can produce similar symptoms. That situation brings greater difficulty in selecting those who effectively have GERD and can benefit from surgery, which requires a more careful preoperative workup (8), as well as the participation of other specialists to decide for the best treatment for each patient.
This paper focuses on how to obtain a successful laparoscopic antireflux surgery in patients with extraesophageal symptoms.
Workup
A panel of expert gastroenterologists and surgeons suggested that every patient who will undergo an antireflux surgery should perform a symptomatic evaluation, barium swallow, upper endoscopy, esophageal manometry, and ambulatory pH monitoring (9). A detailed work up is necessary since many studies have shown that the presence of esophageal symptoms such as heartburn and regurgitation are not enough to diagnose GERD, leading to an incorrect diagnosis in 30% to 50% of patients (10,11). This situation is even more difficult in the presence of extraesophageal syndromes that may mimic other diseases and may not coexist with esophageal symptoms in up to 75% of the patients in many series (2,12).
Upper endoscopy
Endoscopy has a low sensitivity and specificity for the diagnosis of GERD based on the finding of esophagitis (13) especially in patients under antacid medication. Even the presence of hiatal hernia and esophagitis was not different in patients with ear, nose and throat (ENT) symptoms in the absence or presence of GERD confirmed by pH monitoring (14).
The major value of endoscopy; however, is to exclude gastric and duodenal diseases and to detect the presence of Barrett’s esophagus which is usually present in 10% to 14% of GERD patients (13).
pH monitoring
Ambulatory 24-h pH monitoring should be routinely performed in the preoperative workup of patients’ suspect of having GERD (13,14). In combination with multichannel intraluminal impedance (MII-pH) or not 24-h pH monitoring provides the best GERD diagnoses and quantification, as well as allows a temporal correlation between symptoms and episodes of reflux (15). However, while ambulatory pH monitoring is still the most reliable diagnostic method for establishing a diagnosis of GERD, with well-established reference values for distal reflux, it is still unclear the threshold for proximal reflux (16).
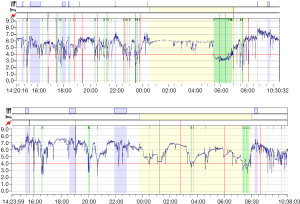
The absence of a wildly accepted reference value for proximal reflux may be explained by individual tissue resistance and visceral sensitivity. A temporal correlation between symptoms and episodes of reflux may indirectly account for these variables (Figure 1). It must also be remembered, though that some symptoms may be incited by the immediate presence of noxious refluxate in the target organ, such as in cough, in others, such as hoarseness due to laryngitis, symptoms reflects a chronic vocal cord injury and there is no direct temporal relation between symptoms and reflux.
Laryngoscopy
Laryngoscopy may be added to the armamentarium of diagnostic tests of extraesophageal symptoms; however, a low positive predictive value for the diagnosis of GERD is anticipated. Laryngoscopic findings suggestive of GERD are not pathognomonic (Figure 2). It is known that healthy subjects can display many of the same signs on laryngoscopy attributed to reflux, with a high degree of interobserver variability in rating laryngoscopic findings. This suggests that laryngoscopy is not a specific test for diagnosing esophagopharyngeal reflux (17).
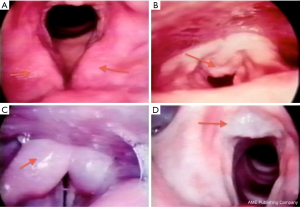
Studies over time (14,18,19) have shown that the laryngoscopic diagnosis of GERD is confirmed at rates around 40% when compared to the results of ambulatory 24-h pH monitoring alone or in combination with MII-pH.
Empiric trial
Empiric trial may be used to help establish GERD as the real cause for symptoms. The response to antacid therapy may indicate refluxed acid as the causative for presented symptoms.
Twice daily proton pump inhibitor (PPI) for 2–3 months is the recommendation (20). Double dose of PPI for a prolonged time is necessary since symptoms may be caused by chronic inflammation that may need extended time for its resolution. Patients unresponsive to this trial should be carefully studied with more specific tests to make a proper diagnosis.
Other tests
Barium esophagogram, as upper endoscopy, may show gastric reflux to the upper esophagus or even pharynx; however, the sensitivity of this test is poor and no objective parameters can be measured. Besides that, the presence of reflux or hiatal hernia on esophagogram did not correlate with reflux on pH monitoring (13) which also shows its low specificity.
Recently, new diagnosis methods have been developed in order to detect the presence of esophagogastric contents in the oropharynx and bronchus. Further studies are still needed to evaluate the real possibility of its clinical application. Among these should be mentioned the system for measurement of aerosolized droplets of acid in the oropharynx (21) and the detection of pepsin and bile acid in the saliva or bronchiolar lavage (22).
Other medical specialists
Symptoms may be caused by diseases of the target organs other than GERD. The consultation of other medical experts, such as pulmonologists, ENT, allergists, dentists, etc. may be necessary to evaluate these patients.
Specific symptoms
Cough
GERD may lead to cough due to: (I) microaspiration of gastric contents to the upper respiratory system; (II) bronchial reflex due to esophageal chemical stimulation; and (III) as a consequence to upper respiratory system chronic inflammation.
pH monitoring may demonstrate a temporal correlation between reflux and cough in case of microaspiration. A positive correlation predicts a good response to surgical therapy in these cases. Patti et al. [2000] (23) showed that cough resolved in 83% of patients in whom a correlation between cough and reflux was found during pH monitoring, but in only 57% of patients when this correlation was absent. Patients that did not present with symptoms during the test had a 0% resolution of cough in the same study.
Cough due to bronchial reflex may be incited by distal esophageal stimulation (24), thus proximal reflux may be absent and a temporal correlation with reflux may be unreliable. In the presence of laryngitis and cough a similar situation may be found. In these patients, a complete work up and the consultation of ENT surgeons are mandatory.
Iqbal et al. (25)—in a systematic review of 13 studies—showed a 60–100% improvement in cough after antireflux surgery.
Laryngitis
GERD is a common cause for laryngitis. Different from cough, laryngitis is represented by a chronic inflammation that may persist even after reflux cessation. Thus, the presence of proximal reflux at the pH monitoring may not be necessary present during the test. Temporal correlation between symptoms of laryngitis (hoarseness, sore throat, etc.) and reflux may not be present for the same reason. Direct evaluation of the larynx (laryngoscopy) may show typical findings to suggest reflux, although with a low accuracy (14).
Few studies focused on the outcomes of laryngeal symptoms after anti-reflux surgery have demonstrated good results in short and middle-term follow-up. Symptomatic improvement ranges from 69% to 97% (26-29). Sala et al. (29) showed superior benefits of surgical therapy in patients with laryngeal symptoms responsive to antacid treatment. These studies pointed out better outcomes after operation in patients without some anatomical abnormalities at the laryngoscopy (such as vocal-cord scarring, paresis, granuloma, and carcinoma or subglottic stenosis) and response to PPI treatment.
Asthma
Similarly to cough, asthma symptoms are caused by microaspiration of gastric contents to the upper respiratory system or bronchial reflex due to esophageal chemical stimulation. Adult-onset asthma is more prone to be caused by GERD (30).
pH monitoring may show reflux but asthma exacerbation will hardly be present during the test to prove GERD as the cause of the disease.
An approach to patients with asthma (with or without concomitant heartburn or regurgitation) is similar to that for patients with chronic cough and laryngitis. A complete workup is necessary and an empiric trial of once- or twice-daily PPIs for 2 to 3 months may be useful (25).
Iqbal et al. (25) reviewed studies totalizing 350 asthma patients observed 49–84% improvement of symptoms after surgery. Field et al. (31) in another review that included 417 asthma patients showed that antireflux surgery improved GERD symptoms, asthma symptoms, asthma medication use, and pulmonary function in 90%, 79%, 88%, and 27%, respectively. Ekstrom et al. (32) in a randomized controlled trial, showed that 12 of 16 patients (75%) had a sustained improvement after antireflux surgery, compared to 9.1% in patients treated medically and 4.2% in the control group.
Dental erosion
Dental erosion due to direct contact of the teeth with refluxed acid leads to characteristic findings of enamel decay.
Although there are no protocols for the investigation of dental erosion due to GERD, we can infer that the same sequence of tests and clinical trial established for other extraesophageal manifestations of GERD should be followed.
There are no data in the literature regarding the outcome of antireflux surgery in the oral repercussions of GERD.
Conclusions
A successful laparoscopic antireflux surgery in patients with extraesophageal symptoms depends on the answer of three questions (Figure 3):
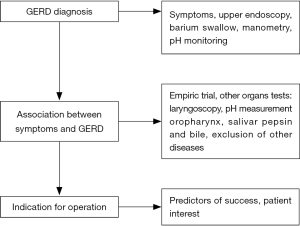
- Does the patient have GERD? An objective test for GERD is mandatory in these patients. The diagnosis of pathological distal reflux is straightforward; however, the diagnosis of proximal reflux and to the target organs may be hard to be established.
- Are the symptoms attributable to GERD? Extraesophageal symptoms may have a different cause than GERD even in the presence of GERD. A rational and complete workup is necessary to answer this question. A single test is not able to correlate symptoms to GERD. Judicious clinical decision based on a sum of different pieces of information is needed.
- Is surgery the appropriate treatment? The correct selection of patients based on predictors for good outcomes shows those who would benefit from surgery. Adequate surgical technical principles should be followed.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, Annals of Laparoscopic and Endoscopic Surgery for the series “Secrets for Successful Laparoscopic Antireflux Surgery”. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/ales.2017.02.06). The series “Secrets for Successful Laparoscopic Antireflux Surgery” was commissioned by the editorial office without any funding or sponsorship. Herbella FA served as the unpaid Guest Editor of the series. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Vakil N, van Zanten SV, Kahrilas P, et al. The Montreal definition and classification of gastroesophageal reflux disease: a global evidence-based consensus. Am J Gastroenterol 2006;101:1900-20; quiz 1943.
- Ing AJ, Ngu MC, Breslin AB. Pathogenesis of chronic persistent cough associated with gastroesophageal reflux. Am J Respir Crit Care Med 1994;149:160-7. [Crossref] [PubMed]
- Frye JW, Vaezi MF. Extraesophageal GERD. Gastroenterol Clin North Am 2008;37:845-58. ix. [Crossref] [PubMed]
- Kollarik M, Ru F, Undem BJ. Acid-sensitive vagal sensory pathways and cough. Pulm Pharmacol Ther 2007;20:402-11. [Crossref] [PubMed]
- Stanghellini V. Relationship between upper gastrointestinal symptoms and lifestyle, psychosocial factors and comorbidity in the general population: results from the Domestic/International Gastroenterology Surveillance Study (DIGEST). Scand J Gastroenterol Suppl 1999;231:29-37. [PubMed]
- Poe RH, Kallay MC. Chronic cough and gastroesophageal reflux disease: experience with specific therapy for diagnosis and treatment. Chest 2003;123:679-84. [Crossref] [PubMed]
- Morgenthal CB, Lin E, Shane MD, et al. Who will fail laparoscopic Nissen fundoplication? Preoperative prediction of long-term outcomes. Surg Endosc 2007;21:1978-84. [Crossref] [PubMed]
- Sidhwa F, Moore A, Alligood E, et al. Diagnosis and Treatment of the Extraesophageal Manifestations of Gastroesophageal Reflux Disease. Ann Surg 2017;265:63-7. [Crossref] [PubMed]
- Jobe BA, Richter JE, Hoppo T, et al. Preoperative diagnostic workup before antireflux surgery: an evidence and experience-based consensus of the Esophageal Diagnostic Advisory Panel. J Am Coll Surg 2013;217:586-97. [Crossref] [PubMed]
- Csendes A, Rencoret G, Beltran M, et al. Relationship between gastroesophageal reflux symptoms and 24 h esophageal pH measurements in patients with normal or minimally abnormal upper endoscopies. Rev Med Chil 2004;132:19-25. [PubMed]
- Chan K, Liu G, Miller L, et al. Lack of correlation between a self-administered subjective GERD questionnaire and pathologic GERD diagnosed by esophageal pH monitoring. J Gastrointest Surg 2010;14:427-36. [Crossref] [PubMed]
- Laukka MA, Cameron AJ, Schei AJ. Gastroesophageal reflux and chronic cough: which comes first? J Clin Gastroenterol 1994;19:100-4. [Crossref] [PubMed]
- Bello B, Zoccali M, Gullo R, et al. Gastroesophageal reflux disease and antireflux surgery-what is the proper preoperative work-up? J Gastrointest Surg 2013;17:14-20; discussion p. 20.
- Herbella FA, Andolfi C, Vigneswaran Y, et al. Importance of esophageal manometry and pH monitoring for the evaluation of otorhinolaryngologic (ENT) manifestations of GERD. A multicenter study. J Gastrointest Surg 2016;20:1673-8. [Crossref] [PubMed]
- Gawron AJ, Hirano I. Advances in diagnostic testing for gastroesophageal reflux disease. World J Gastroenterol 2010;16:3750-6. [Crossref] [PubMed]
- Merati AL, Lim HJ, Ulualp SO, et al. Meta-analysis of upper probe measurements in normal subjects and patients with laryngopharyngeal reflux. Ann Otol Rhinol Laryngol 2005;114:177-82. [Crossref] [PubMed]
- Hicks DM, Ours TM, Abelson TI, et al. The prevalence of hypopharynx findings associated with gastroesophageal reflux in normal volunteers. J Voice 2002;16:564-79. [Crossref] [PubMed]
- de Bortoli N, Nacci A, Savarino E, et al. How many cases of laryngopharyngeal reflux suspected by laryngoscopy are gastroesophageal reflux disease-related? World J Gastroenterol 2012;18:4363-70. [Crossref] [PubMed]
- Hayat JO, Yazaki E, Moore AT, et al. Objective detection of esophagopharyngeal reflux in patients with hoarseness and endoscopic signs of laryngeal inflammation. J Clin Gastroenterol 2014;48:318-27. [Crossref] [PubMed]
- Tsoukali E, Sifrim D. Investigation of extraesophageal gastroesophageal reflux disease. Ann Gastroenterol 2013;26:290-5. [PubMed]
- Hom C, Vaezi MF. Extraesophageal manifestations of gastroesophageal reflux disease. Gastroenterol Clin North Am 2013;42:71-91. [Crossref] [PubMed]
- Wassenaar E, Johnston N, Merati A, et al. Pepsin detection in patients with laryngopharyngeal reflux before and after fundoplication. Surg Endosc 2011;25:3870-6. [Crossref] [PubMed]
- Patti MG, Arcerito M, Tamburini A, et al. Effect of laparoscopic fundoplication on gastroesophageal reflux disease-induced respiratory symptoms. J Gastrointest Surg 2000;4:143-9. [Crossref] [PubMed]
- Rosztóczy A, Makk L, Izbéki F, et al. Asthma and gastroesophageal reflux: clinical evaluation of esophago-bronchial reflex and proximal reflux. Digestion 2008;77:218-24. [Crossref] [PubMed]
- Iqbal M, Batch AJ, Spychal RT, et al. Outcome of surgical fundoplication for extraesophageal (atypical) manifestations of gastroesophageal reflux disease in adults: a systematic review. J Laparoendosc Adv Surg Tech A 2008;18:789-96. [Crossref] [PubMed]
- Oelschlager BK, Quiroga E, Parra JD, et al. Long-term outcomes after laparoscopic antireflux surgery. Am J Gastroenterol 2008;103:280-7; quiz 288. [Crossref] [PubMed]
- Westcott CJ, Hopkins MB, Bach K, et al. Fundoplication for laryngopharyngeal reflux disease. J Am Coll Surg 2004;199:23-30. [Crossref] [PubMed]
- Catania RA, Kavic SM, Roth JS, et al. Laparoscopic Nissen fundoplication effectively relieves symptoms in patients with laryngopharyngeal reflux. J Gastrointest Surg 2007;11:1579-87; discussion 1587-8. [Crossref] [PubMed]
- Sala E, Salminen P, Simberg S, et al. Laryngopharyngeal reflux disease treated with laparoscopic fundoplication. Dig Dis Sci 2008;53:2397-404. [Crossref] [PubMed]
- Harding SM. Acid reflux and asthma. Curr Opin Pulm Med 2003;9:42-5. [Crossref] [PubMed]
- Field SK, Gelfand GA, McFadden SD. The effects of antireflux surgery on asthmatics with gastroesophageal reflux. Chest 1999;116:766-74. [Crossref] [PubMed]
- Ekström T, Johansson KE. Effects of antireflux surgery on chronic cough and asthma in patients with gastroesophageal reflux disease. Respir Med 2000;94:1166-70. [Crossref] [PubMed]
Cite this article as: Laurino Neto RM, Herbella FAM, Pinna BR, Patti MG. Secrets for a successful laparoscopic antireflux surgery: patients with extraesophageal symptoms. Ann Laparosc Endosc Surg 2017;2:26.


